In a new study, published in Nature, AII researchers and collaborators from the Vrije Universiteit and CSSB Hamburg reveal the structural basis of an intact type VII secretion system inner membrane machinery from Mycobacterium tuberculosis. Type VII secretion systems are molecular machines which play key roles in the infection cycle of many pathogenic mycobacteria, including the notorious Mycobacterium tuberculosis. A deeper understanding on the structure and function of these systems can enable the development of novel therapies for the treatment tuberculosis.
Prior to the corona virus pandemic, tuberculosis was the leading cause of death worldwide from a single infectious agent. Mistakenly considered by many to be a disease of the past, tuberculosis is a disease that still kills four thousand people every day1. With the spotlight placed on combatting COVID-19, the fight against tuberculosis has now suffered additional difficulties, due to decreased options for testing and treatment during lockdown periods. WHO models are currently estimating an excess of half a million TB deaths in 2020 and a decade’s worth setback in the fight against TB2.
To initiate infection, bacterial proteins and other nutrients must travel across the mycobacteria’s distinct impermeable double membrane. “Mycobacterium tuberculosis possesses a dedicated arsenal of molecular machineries that facilitate this transport,” explains Wilbert Bitter from the AII-Amsterdam UMC and Vrije Universiteit Amsterdam “and this pathogen has five different type VII secretion systems (T7SS) that are not only central for virulence but also critical for nutrient uptake, making it an interesting drug target.”
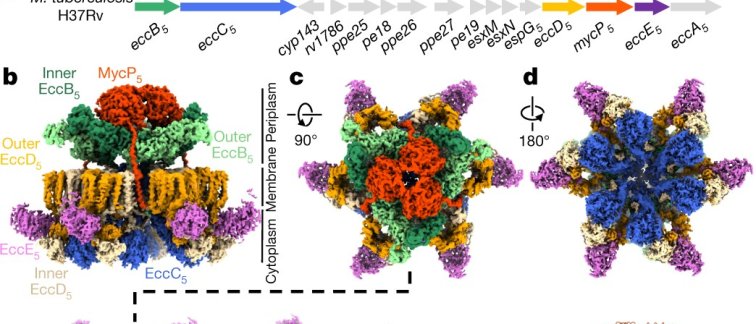
Previous studies conducted by the researchers revealed that four T7SSs components form six-sided star shaped complexes, however high-resolution structural information and mechanistic insights could not be determined. “We are now able to see that this large molecular machine has a staggering 165 membrane anchors”, notes investigator Edith Houben from the Vrije Universiteit Amsterdam, “and we can understand how the components are kept together, as one stable assembly”. The researchers focused their efforts on the nanomachinery that crosses the inner membrane of Mycobacterium tuberculosis. To obtain large quantities of proteins and bypass the high infectivity of pathogenic mycobacteria, VUmc researcher Roy Ummels reconstituted the systems in non-pathogenic mycobacterial species from which cryo-EM samples were purified. After many trials to obtain a sample that is suitable for cryo-EM imaging, high-resolution information was collected at CSSB’s cryo-electron microscopy facility. “It was a very exciting journey to see how the quality of the samples has improved over the years to the point where we were impatient for the microscope to be available to us” notes CSSB researcher Jiri Wald.
Cryo-EM analysis not only confirmed the star-like assembly, but also revealed how this complex is stabilized by an additional fifth component, the MycP5 protease, which is essential for the function of the T7SS. The structural model shows a group of three MycP5 enzymes capping a dome-like chamber at the top of the complex. Each MycP5 sits on top, like an inverted cherry, and provides stability by increasing the number of contact points among subunits of the complex. “In the absence of MycP5 the entire complex becomes more flexible and the arms of the hexameric star begin to wobble”, explains CSSB researcher and first author Catalin Bunduc “The complex with MycP5 is like a stable suspension bridge and when the suspenders (MycP5) are removed, the complex lacks support and sways like a wobbly footbridge.”
More often, protein transport happens in an unfolded state, meaning that prior to traveling through a secretion channel the protein is untangled into a chain-like structure, reducing its width. T7SS, however, secretes proteins in a folded manner thus requiring a relatively large opening in the bacterial membrane. The data provided new insights into how T7SSs could be able to secrete these large, folded proteins in a controlled manner without allowing other molecules to leak in. “The membrane anchors of the motor protein assemble into bundles that seal the central secretion channel” notes CSSB researcher Dirk Fahrenkamp, “And the machinery seems to form two communicating chambers upon active secretion, potentially preventing leakage when the pore is opened”.
While tuberculosis can be treated with antibiotics, drug resistant strains are becoming increasingly more prevalent and in 2019 these represented 3.5% of new TB cases and 17.7% of previously treated cases. The structural insights gained by the researchers provide a starting platform for the identification of domains/interactions that could be targets for drug development. “Our results are a huge step forward in understanding T7SSs and Mycobacterium tuberculosis itself” explain the authors “we can now investigate potential drug binding sites that inhibit the function of T7SS and prevent the propagation of infection.”
“This breakthrough not only reveals new possibilities in the fight against tuberculosis but also emphasizes the societal importance of fundamental research. Breakthroughs, like this one, can ultimately lead to practical applications that save lives,” marks CSSB investigator Thomas Marlovits.
Link to article Structure and dynamics of a mycobacterial type VII secretion system | Nature