The 28th of February is Rare Disease Day, an important day to raise awareness for patients, families, and caregivers affected by rare diseases. A disease is considered rare when it affects fewer than 1 in 2000 people. But added up, worldwide, more than 300 million people have a rare disease, and over 70% of them are genetic in origin. Despite their prevalence, finding effective treatments remains a challenge.Smarter Use of Medication Needed for Chronic Bowel Diseases
Rethinking Drug Development: The Role of Academia
Rare diseases affect small patient populations, and since pharmaceutical companies often see limited financial incentive to invest in new therapies, there is a greater role for academia here. At Amsterdam UMC, the initiative ‘Medicijn voor de Maatschappij’ (Medicine for Society, MvdM) is leading the way in developing and making therapies for rare diseases more accessible. AGEM PhD researcher, Sibren van den Berg, coordinates various MvdM projects, focusing on a new approach: “The goal is to prevent problems in the availability of medicines by working differently right from the start,” Sibren explains.
This means involving researchers early and encouraging them to consider not just the science, but also sustainable availability, regulations, and socially responsible pricing. By thinking ahead, the team aims to ensure that new therapies don’t just get developed but also reach the patients who need them by taking into account affordability.
Success Stories: Repurposing Old Drugs for New Hope
This approach is showing results. For example, in collaboration with Amsterdam UMC and other partners, MvdM has successfully repurposed existing drugs for rare diseases. One such example is Fenformine, a cousin of metformin, originally used for diabetes patients but withdrawn from the market since it resulted in high risk for lactic acidosis. Nevertheless, it showed potential in laboratory studies for pancreatic cancer patients. Thanks to the collaboration between academic centers, patient foundations, and MvdM, a new company was set up to further develop and test this drug in a socially responsible way. The aim is not only to bring the medicine to market, but also to keep it affordable and accessible for patients.
Gene therapy to treat Rare Diseases
Since so many rare diseases are caused by mutations in a single gene, another promising therapeutic option is gene editing to correct the root cause rather than just managing symptoms. Developing scalable and safe gene editing for rare diseases is the main focus of AGEM researchers postdoctoral researcher Mouraya Hussein and assistant professor Daniël Warmerdam.
Gene editing allows for the correction of disease-causing mutations via molecular scissors like CRISPR-Cas9. Mourayais particularly excited about prime editing, a next-generation gene editing approach, that use a modified enzyme to “nick” just one strand of DNA, rather than cutting both.
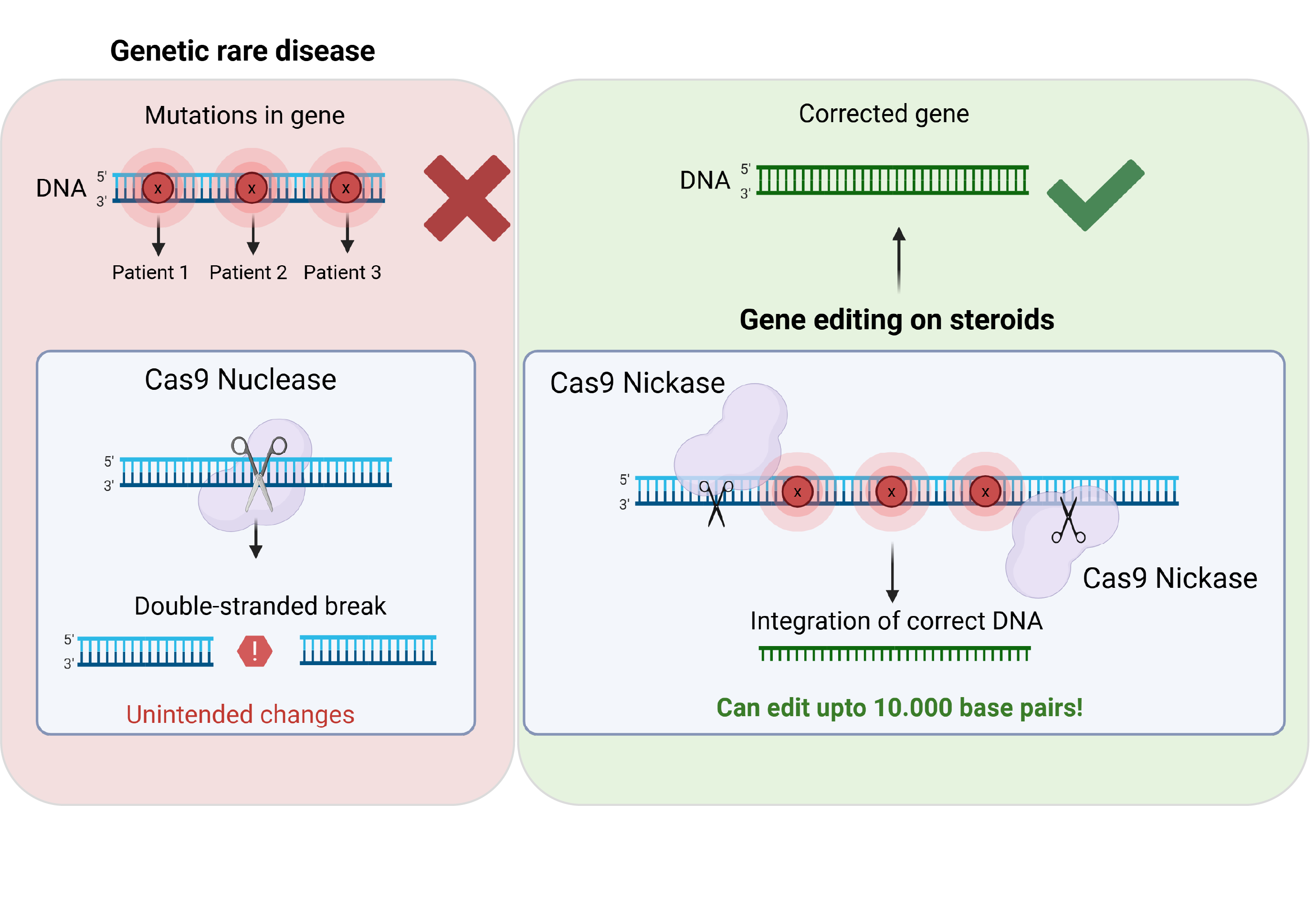 Cas9 gene editing (left) vs 'prime editing on steroids' (right)
Cas9 gene editing (left) vs 'prime editing on steroids' (right)
This more controlled mechanism allows for accurate correction of disease-causing mutations while reducing unintended changes. This gentler alternative, especially in combination with a recombinase enzyme, also referred to by Mouraya as ‘prime editing on steroids’, allows for editing relatively large DNA parts up to 10.000 base pairs.
For rare genetic diseases, where each patient might have a unique mutation, this technology could enable personalized corrections for a wider group of patients within a single disease.
While ‘prime editing on steroids’ holds great promise, Daniel emphasizes the importance of understanding the safety and off-target effects of these gene therapies. Unfortunately, although computations models can predict where edits should occur, discrepancies between predicted and actual editing sites can arise. That is why Mouraya and Daniël explain that the team is systematically testing these strategies in human cell lines, including patient-derived induced pluripotent stems cells (iPSCs). Working with patient-derived material also helps anticipate potential immune responses and biological variability before therapies reach clinical testing. This careful, stepwise approach is essential to ensure that gene editing therapies are both safe and accessible.
Where do we start?
Once there is more clarity about the off-target effects and safety, the next question is: where can we start? With around 8,000 genetic diseases, deciding where to start is a challenge in itself. A safe and efficient way to administer these gene therapies is via nanoparticles, which we know from the COVID vaccine. As for deciding in which patients to start testing gene therapy; a thing to consider is that this technique is highly efficient at delivering gene editing components to the liver, where these nanoparticles naturally accumulate, says Daniel. This could be particularly useful for rare diseases where the liver plays a key role, such as those involving toxic metabolites produced in the liver which in turn can damage brain cells.
The eye represents another compelling target tissue for gene editing therapies. Inherited retinal diseases are often caused by well-defined genetic mutations, and the eye allows for local administration of therapeutics, thereby limiting systemic exposure and reducing safety risks.
That being said, before we can actually apply these gene therapies in patients, there is a strong need for more functional validation and data, both pre-clinical and clinical. On a positive note, we have many AGEM researchers working together on bringing research into reality and developing feasible treatments for rare diseases.
Authors
Merel Goedkoop
Marte Molenaars